- Mycoplasma Testing
- Staff available on site for training and discussion (cell immortalization, development of primary cell cultures, etc.)
- Cell Line Authentication via the UTMB Molecular Genomic Core
Studies have repeatedly shown that the incidence of mycoplasma contamination ranges from 25% to 87% of cell cultures (G.J. McGarrity and H. Kotani. In: The Mycoplasmas, Vol. IV. S. Razin and M.F. Barile (eds) Academic Press. pp. 353-390, 1985). Mycoplasma
can be introduced into cell cultures by laboratory personnel and into newly introduced contaminated cell lines as well by reagents. Their small size (approximately 0.2 µm) allows them to pass through most 0.22-µm sterilization filters,
and they are resistant to common antibiotics such as penicillin and streptomycin (C. Lincoln and M. Gabridge. Methods
Cell Biol. 57:49-65, 1998).
The TCCF is recognized as being vigilant in monitoring cell culture quality, giving advice to investigators on handling and, importantly, preventing mycoplasma contamination. Investigators are encouraged to use antibiotic-free media to prevent the development
of antibiotic-resistant bacterial and mycoplasma strains and to avoid the effects of antibiotics on cellular metabolism.
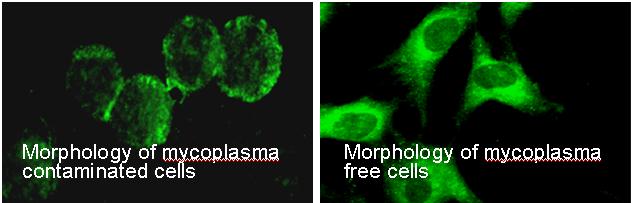
Contamination by mycoplasma species can easily lead to unreliable and unreproducible results and experiments (J Tang, et al. Microbiology Methods. 39: 121-126, 2000).
 Several
methods have been developed to detect mycoplasma contamination.
Several
methods have been developed to detect mycoplasma contamination.
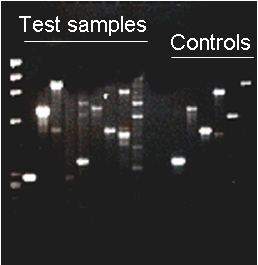
The TCCF uses nested PCR and fluorescent microscopy simultaneously to ensure the best and most accurate results. The primer set used allows detection of various mycoplasma species (M. fermentans, M. hyorhinis, M. arginini,
M. orale, M. salivarium, M. hominis, M. pulmonis, M. arthritidis, M. bovis, M. pneumoniae, M. pirum and M. caprocolum), as well as Acholeplasma and Spiroplasma species, with high sensitivity and specificity (an example is shown in this figure). TCCF also offers mycoplasma testing of culture media and serum and media supplements.
Questions? Please contact Steve Boldogh, Director,
at (409) 772-9414 or Yixiao Sun at (409) 772-4903.